Smell memories in the fly brain and what makes individual animals unique
/Have you ever woken up in the morning to the refreshing aroma wafting from the neighborhood cafe? Does it remind you of the last Christmas Eve sitting with family enjoying a cup of coffee, or the cozy conversations with an old friend in you two’s favorite coffee shop? Smelling an odor can cause an emotional reaction or elicit a memory that is unique to that individual person. Despite smelling the same exact odor, a different individual might have a totally different reaction, shaped by the unique life experiences that individual has had in association with a given fragrance. Neuroscientists call this associative learning. This topic has been studied extensively for decades, because our unique experiences, which shape our brains through mechanisms of learning and memory, define who we are and how we view the world. The olfactory system of fruit flies can also mediate associative learning. And a structure called the mushroom body plays a key role. Thanks to its relatively simple anatomical architecture, it has served as an excellent model system to elucidate common mechanisms underlying olfactory learning. Although previous work beautifully mapped the anatomical structure of this circuit, it has remained elusive how the patterns of neural activity in the mushroom body and its downstream structures serve as a substrate for learning.
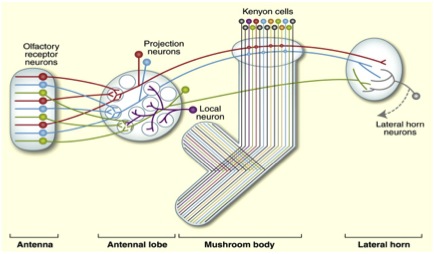
There are steps that are needed for olfactory signals to reach the mushroom body. First, olfactory information enters the brain by being detected by olfactory sensory neurons in the fly’s antenna. Second this information is passed on to the second-order projection neurons, located in the antenna lobe. Third, these second-order projection neurons send this processed olfactory information on to mushroom body neurons called Kenyon Cells (KC’s). The mushroom body lobes also contain the axonal arbors of the more than 2000 Kenyon Cells (KC’s). Here, in the mushroom body, these ~2000 axonal arbors synapse onto the dendrites of only 34 downstream neurons named mushroom body output neurons (MBON’s).
Olfactory Information Flow:
Does this striking convergence in neuronal projections, from more than 2000 KC’s to only 34 MBON’s, result in a transition in signal processing from KC’s to MBON’s? To answer this question, the researchers wanted to see the activity patterns of different MBON’s. So, to visualize these activity patterns they applied a powerful fly genetics tool called UAS-GAL4 system, which enables the researchers to express a target protein in any specific type of cell one is interested in studying. Using this system, they expressed the protein GCaMP, which can generate fluorescence signals when the neurons are activated to visualize the neuronal activity in different types of MBON’s. Based on the neuronal responses of different MBONs to 10 different odor stimuli, they found that these MBON’s are broadly tuned to a wide range of stimuli, and highly correlated in their response patterns. This is in sharp contrast to the response features of KCs which exhibit sparse coding, specific tuning and signal decorrelation. For example, several MBON’s can all respond strongly to both apple cider vinegar and yeast, but one individual KC might respond only to one of them. The researchers concluded that there does exist a pronounced change in olfactory signal processing consistent with the structural convergence from KC’s to MBON’s.
Considering that the fruit fly olfactory system is a model system for studying associative learning, the researchers are also curious about whether these mushroom body neurons show learning-based individualization. Thus they compared the response patterns of a single type of MBON across genetically identical individual flies. Indeed, they discovered high cross-individual variability in response patterns of the same type of MBON: the odors that would activate one cell type in fly A are very different than the odors that would activate this same type in fly B. This finding suggests that individualization in olfactory signal processing occurs in MBONs, which is very likely a result of individual olfactory learning experiences that are unique to each fly and can shape the connection between KC’s and MBON’s.
In search of the functional mechanism underlying olfactory associative learning, the researchers discovered the striking transition in signal processing from KC’s to MBON’s in the fruit fly mushroom body lobes. Moreover, they found high variability and individuality in the response patterns of MBONs between genetically identical fly individuals, which suggests that the neural activity in MBON’s reflects synaptic plasticity resulting from individualized olfactory learning. All these findings from fly olfactory system could find their analogy in vertebrate animals, and therefore shed light on the common and fundamental computational mechanism underlying associative learning.